Welcome to the REPLACE study
Clinical data indicate that 20–60% of patients with PAHPAH, pulmonary arterial hypertension receiving PDE5iPDE5i: phosphodiesterase type 5 inhibitor do not reach or maintain treatment goals. Switching to riociguat may be a treatment option for these patients. The REPLACE study investigated the efficacy and safety of switching from PDE5iPDE5i: phosphodiesterase type 5 inhibitor to riociguat compared with remaining on PDE5iPDE5i: phosphodiesterase type 5 inhibitor in patients with PAHPAH, pulmonary arterial hypertension at intermediate risk.
REPLACE key results
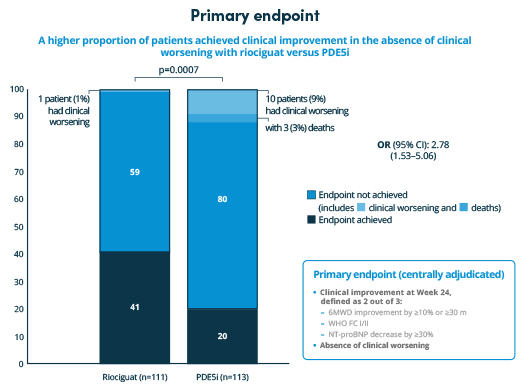
The REPLACE study met its primary endpoint. Switching from PDE5iPDE5i: phosphodiesterase type 5 inhibitor to riociguat resulted in a significantly higher proportion of patients experiencing clinical improvement and significantly lower rates of clinical worsening compared with remaining on PDE5iPDE5i: phosphodiesterase type 5 inhibitor .

Professor Marius Hoeper
presents the REPLACE study results
Resources

Find resources associated with the study, including other riociguat publications